qPCR array - page 1 qPCR array - page 2
Introduction - qPCR arrays qPCR arrays
are the most reliable tools for parallel
quantitative analysis of gene expression
signatures of a focused panel of genes. They are
available for mRNAs and microRNAs expression
profiling in 96-well, 384-well and (hopefully
soon) in 1536-well formats. The detection
chemistry used in qPCR arrays is either SYBR Green
I (or comparable DNA binding dyes) or optimized
probe-based primer
sets (mainly TaqMan
probes or LNA probes).
They
can screen the entire panel of expressed microRNA
or sub-panels, e.g. pathway- or disease-focused
gene families. qPCR arrays can also be customized
to contain a panel of genes tailored to your
specific research interests.
Why using qPCR arrays?
qPCR array performance
Simple workflow
Simply mix your cDNA template with the appropriate ready-to-use PCR master mix, aliquot equal volumes to each well of the same plate, and then run the real-time PCR cycling program. PCR Arrays are compatible with any block based real-time cycling system => http://cyclers.gene-quantification.info/ Well designed plate
layout and multiple controls on various levels
The PCR Arrays are available in both 96-well, 384-well and 1536-well plates and are used to monitor the expression of 84 up to over 1000 genes related to a disease state or pathway plus multiple reference genes. Multiple controls are also included on each array for genomic DNA contamination, RNA quality, and general PCR performance. Following controls are added to the qPCR array:
Easy-to-use
normalisation and expression profiling data
analysis
Data analysis can be done in a simple version on the basis of Excel-based data analysis template, or by the GPR method (see below) or using high complex analysis software tools like Genex (MultiD, Sweden). Data analysis is mainly based on the ΔΔCt method (Livak & Schmittgen, 2001) with normalization of the raw data to either housekeeping genes or an external RNA control. See this nice webinar about real-time PCR data analysis by Prof. Mikael Kubista on "Statistical approaches to gene expression profiling with GenEx 4" The new software versions are available here => http://genex.gene-quantification.info/ qPCR array application papers PCR’s next frontier PCR The workhorse of modern molecular biologyis charging forward using both conventional and digital methods to explore single cells and even single molecules. Nathan Blow reports. NATURE METHODS VOL 4(10) 2007: 869 Reproducibility of quantitative RT-PCR array in miRNA expression profiling and comparison with microarray analysis. Chen Y, Gelfond JA, McManus LM, Shireman PK. Department of Surgery, University of Texas Health Science Center, San Antonio, TX 78229, USA BMC Genomics. 2009 Aug 28;10: 407. BACKGROUND:
MicroRNAs (miRNAs) have critical functions in
various biological processes. MiRNA profiling is
an important tool for the dentification of
differentially expressed miRNAs in normal cellular
and disease processes. A technical challenge
remains for high-throughput miRNA expression
analysis as the number of miRNAs continues to
increase with in silico prediction and
experimental verification. Our study critically
evaluated the performance of a novel miRNA
expression profiling approach, quantitative RT-PCR
array (qPCR-array), compared to miRNA detection
with oligonucleotide microchip (microarray).
RESULTS: High
reproducibility with qPCR-array was demonstrated
by comparing replicate results from the same RNA
sample. Pre-amplification of the miRNA cDNA
improved sensitivity of the qPCR-array and
increased the number of detectable miRNAs.
Furthermore, the relative expression levels of
miRNAs were maintained after pre-amplification.
When the performance of qPCR-array and microarrays
were compared using different aliquots of the same
RNA, a low correlation between the two methods
(r=-0.443) indicated considerable variability
between the two assay platforms. Higher variation
between replicates was observed in miRNAs with low
expression in both assays. Finally, a higher false
positive rate of differential miRNA expression was
observed using the microarray compared to the
qPCR-array.
CONCLUSION:
Our studies demonstrated high reproducibility of
TaqMan qPCR-array. Comparison between different
reverse transcription reactions and qPCR-arrays
performed on different days indicated that reverse
transcription reactions did not introduce
significant variation in the results. The use of
cDNA pre-amplification increased the sensitivity
of miRNA detection. Although there was
variability
associated with pre-amplification in low abundance
miRNAs, the latter did not involve any systemic
bias in the estimation of miRNA expression.
Comparison between microarray and qPCR-array
indicated superior sensitivity and specificity of
qPCR-array.
Customized Molecular Phenotyping by Quantitative Gene Expression and Pattern Recognition Analysis Shreeram Akilesh, Daniel J. Shaffer, and Derry Roopenian Genome Res. 2003 13(7): 1719-1727 The Jackson Laboratory, Bar Harbor, Maine 04609, USA Description
of the molecular phenotypes of pathobiological
processes in vivo is a pressing need in genomic
biology.We have implemented a high-throughput
real-time PCR strategy to establish quantitative
expression profiles of a customized set of target
genes.It enables rapid, reproducible data
acquisition from limited quantities of RNA,
permitting serial sampling of mouse blood during
disease progression.We developed an easy to use
statistical algorithm—Global Pattern
Recognition—to readily identify genes whose
expression has changed significantly from healthy
baseline profiles.This approach provides unique
molecular signatures for rheumatoid arthritis,
systemic lupus erythematosus, and graft versus
host disease, and can also be applied to defining
the molecular phenotype of a variety of other
normal and pathological processes.
Activated NKT Cells Inhibit Autoimmune Diabetes through Tolerogenic Recruitment of Dendritic Cells to Pancreatic Lymph Nodes Yi-Guang Chen, Caroline-Morgane Choisy-Rossi, Thomas M. Holl, Harold D. Chapman, Gurdyal S. Besra, Steven A. Porcelli, Daniel J. Shaffer, Derry Roopenian, S. Brian Wilson, and David V. Serreze The Journal of Immunology, 2005, 174: 1196-1204 This paper
tried to determine why a drug (a-GalCer or
a-galactosylceramide) could inhibit the onset of
autoimmune diabetes. Among many experiments,
GPR was used to determine why a systemic drug
(a-GalCer) treatment elicited the migration of
mature Dendritic Cells (DC) and T cells into the
Pancreatic Lymph Nodes (PLNs), but not the
Mesenteric Lymph Nodes (MLNs) of Non-Obese
Diabetic (NOD) mice. They compared the expression
levels by real-time PCR of genes encoding 19
different chemokines and 11 of their receptors in
the PLNs and MLNs of NOD mice that were either
untreated or injected 24 h previously with
a-GalCer. There were no significant
differences between PLNs and MLNs from untreated
mice. However, CCL17 (5.3-fold), CCL19 (2.4-fold),
CCL5 (1.8-fold), and CXCL16 (1.6-fold) gene
expression levels were significantly higher in the
PLNs than MLNs of a-GalCer-treated NOD mice.
Conversely, CCR4 (3.3-fold) and CCR6 (1.7-fold)
were expressed at higher levels in the MLNs of the
a-GalCer-treated NOD mice. This differential level
of chemokine expression may underlie the migration
of DCs and T cells to the PLNs, but not the MLNs,
of a-GalCer-treated NOD mice thus resulting in an
inhibition of autoimmune diabetes.
A Real-Time PCR Array for Hierarchical Identification of Francisella Isolates Kerstin Svensson1,2, Malin Granberg1, Linda Karlsson1, Vera Neubauerova3, Mats Forsman1, Anders Johansson1,2 1 Division of CBRN Defense and Security, Swedish Defense Research Agency, Umeå, Sweden, 2 Department of Clinical Microbiology, Infectious Diseases and Bacteriology, Umeå University, Umeå, Sweden, 3 Central Military Health Institute, Prague, Czech Republic PLoS ONE 4(12): e8360 A robust,
rapid and flexible real-time PCR assay for
hierarchical genetic typing of clinical and
environmental isolates of Francisella is
presented. Typing markers were found by multiple
genome and gene comparisons, from which 23
canonical single nucleotide polymorphisms
(canSNPs) and 11 canonical insertion-deletion
mutations (canINDELs) were selected to provide
phylogenetic guidelines for classification from
genus to isolate level. The specificity of the
developed assay, which uses 68 wells of a 96-well
real-time PCR format with a detection limit of 100
pg DNA, was assessed using 62 Francisella isolates
of diverse genetic and geographical origins. It
was then successfully used for typing 14 F.
tularensis subsp. holarctica isolates obtained
from tularemia patients in Sweden in 2008 and five
more genetically diverse Francisella isolates of
global origins. When applied to human ulcer
specimens for direct pathogen detection the
results were incomplete due to scarcity of DNA,
but sufficient markers were identified to detect
fine-resolution differences among F. tularensis
subsp. holarctica isolates causing infection in
the patients. In contrast to other real-time PCR
assays for Francisella, which are typically
designed for specific detection of a species,
subspecies, or strain, this type of assay can be
easily tailored to provide appropriate
phylogenetic and/or geographical resolution to
meet the objectives of the analysis.
The nonhomologous end joining factor Artemis suppresses multi-tissue tumor formation and prevents loss of heterozygosity Y Woo, SM Wright, SA Maas1,7, TL Alley1, LB Caddle1, S Kamdar1, J Affourtit, O Foreman1, EC Akeson, D Shaffer, RT Bronson, HC Morse, D Roopenian and KD Mills Oncogene. 2007 26(41): 6010-6020 Used an early
version of the StellARray to confirm CGH
(Comparative Genome Hybridization) results in a
mouse model to cancer however they did not publish
their use of GPR but instead used classical
statistical measures.Nonhomologous
end joining (NHEJ) is a critical DNA repair
pathway, with proposed tumor suppression functions
in many tissues. Mutations in the NHEJ factor
ARTEMIS cause radiation-sensitive severe combined
immunodeficiency in humans and may increase
susceptibility to lymphoma in some settings. We
now report that deficiency for Artemis (encoded by
Dclre1c/Art in mouse) accelerates tumorigenesis in
several tissues in a Trp53 heterozygous setting,
revealing tumor suppression roles for NHEJ in
lymphoid and non-lymphoid cells. We also show that
B-lineage lymphomas in these mice undergo loss of
Trp53 heterozygosity by allele replacement, but
arise by mechanisms distinct from those in Art
Trp53 double null mice. These findings demonstrate
a general tumor suppression function for NHEJ, and
reveal that interplay between NHEJ and Trp53 loss
of heterozygosity influences the sequence of
multi-hit oncogenesis. We present a model where
p53 status at the time of tumor initiation is a
key determinant of subsequent oncogenic
mechanisms. Because Art deficient mice represent a
model for radiation-sensitive severe combined
immunodeficiency, our findings suggest that these
patients may be at risk for both lymphoid and
non-lymphoid cancers.
General Normalistion Stategies in real-time PCR Real-Time PCR:
Current Technology and Applications
Publisher: Caister Academic Press Editor: Julie Logan, Kirstin Edwards and Nick Saunders Applied and Functional Genomics, Health Protection Agency, London (2009) ISBN: 978-1-904455-39-4 http://www.horizonpress.com/realtimePCR Chapter 4 - Reference Gene Validation Software for Improved Normalization J. Vandesompele, M. Kubista and M. W. Pfaffl (2009) Real-time
PCR is the method of choice for expression
analysis of a limited number of genes. The
measured gene expression variation between
subjects is the sum of the true biological
variation and several confounding factors
resulting in non-specific variation. The purpose
of normalization is to remove the non-biological
variation as much as possible. Several
normalization strategies have been proposed, but
the use of one or more reference genes is
currently the preferred way of normalization.
While these reference genes constitute the best
possible normalizers, a major problem is that
these genes have no constant expression under
all experimental conditions. The experimenter
therefore needs to carefully assess whether a
certain reference gene is stably expressed in
the experimental system under study. This is not
trivial and represents a circular problem.
Fortunately, several algorithms and freely
available software have been developed to
address this problem. This chapter aims to
provide an overview of the different concepts.
Chapter 5 - Data Analysis Software M. W. Pfaffl, J. Vandesompele and M. Kubista (2009) Quantitative
real-time RT-PCR (qRT-PCR) is widely and
increasingly used in any kind of mRNA
quantification, because of its high sensitivity,
good reproducibility and wide dynamic
quantification range. While qRT-PCR has a
tremendous potential for analytical and
quantitative applications, a comprehensive
understanding of its underlying principles is
important. Beside the classical RT-PCR
parameters, e.g. primer design, RNA quality, RT
and polymerase performances, the fidelity of the
quantification process is highly dependent on a
valid data analysis. This review will cover all
aspects of data acquisition (trueness,
reproducibility, and robustness), potentials in
data modification and will focus particularly on
relative quantification methods. Furthermore
useful bioinformatical, biostatical as well as
multi-dimensional expression software tools will
be presented.
Real-Time
PCR: Current Technology and Applications
- Book reviews:
Companies providing qPCR arrays:
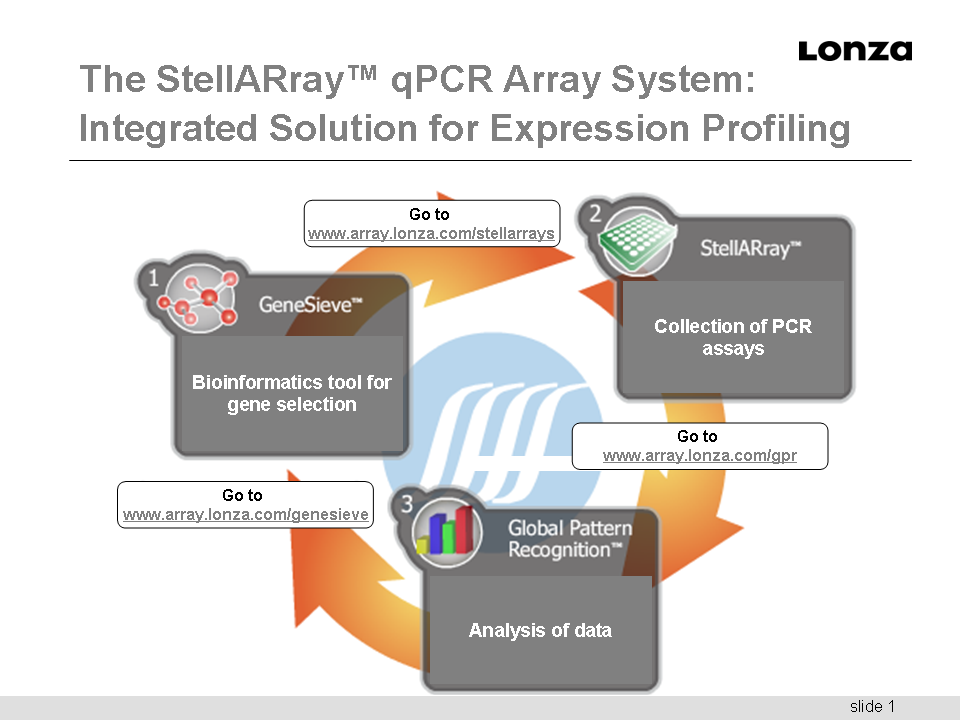
LONZA & Bar Harbor Biotechnology White Paper: StellARray Gene Expression System - Revealing Profiles with Unbiased Significance Daniel Shaffer, Aaron Brown, William Olver Bar Harbor BioTechnology, Inc. and Marjorie Smithhisler, Lonza Walkersville, Inc. In this
paper, we present three application ex-amples
demonstrating the utility of the StellARray Gene
Expression System to reveal gene expression level
changes in diverse biological contexts such as
toxicology, cancer, and stem cell
differentiation. By combining Clonetics and
Poietics Primary Human Cells with the StellARray
Gene Expression System, all from Lonza, the
researcher is pro-vided with a synergistic system
to reveal gross and subtle changes in gene
expression when analyzing in vitro models of human
tissues. This is accomplished easily in 96-
and 384-well formatted StellARrayqPCR Arrays using
a stan-dard qPCR instrument and a generic SYBR®
Green-based Reagent Master Mix. The Global Pattern
Recognition (GPR) Data Analysis Tool is
optimally suited to generate a ranked list of
significantly changed genes within a qPCR dataset.
GPR overcomes the inconsistencies associated with
con-ventional single gene normalization procedures
by eliminat-ing a priori normalizer selection.
Overall, the results show how the StellARray Gene
Expression System eliminates false positives and
provides TRUE results that are backed by a
rigorous statistical analysis.
Simple and accurate analysis of Real-Time PCR data using Bar Harbor Biotechnology GPR software http://www.bhbio.com/products/gpr/ Bar Harbor Biotechnology has solved one of the most fundamental problems facing experimentation using Real-Time PCR. How do I analyze the data and determine REAL changes in gene expression? The answer to this question is found in Bar Harbor Biotechnology, Inc.'s patent pending Global Pattern Recognition (GPR) algorithm, which makes gene expression analysis simple, fast and reliable. Here are some reasons why we developed this algorithm. Real-time dogma #1 - using single gene
normalizers
The traditional approach to measure gene expression changes from Real-Time PCR data has been to normalize the results of a gene of interest with respect to a housekeeping gene (aka. a reference or normalizer gene). The general assumption is that the level of expression of the normalizer gene does not change in the context of the experiment and can be used to normalize the variability in RNA quantity between individual samples. By normalizing to a housekeeping gene, in theory, a magnitude of change can be calculated between groups of samples for a gene of interest. However, this mode of analysis is greatly complicated by the fact that housekeeping genes commonly used as normalizers (e.g., GAPDH, β-actin, and HPRT) themselves can change in apparent expression when comparing tissues or cells in different states (Bustin 2000; Schmittgen et al. 2000; Goidin et al. 2001; Hamalainen et al. 2001). 18S rRNA is another normalizer that intuitively and experimentally seems more stable, but even 18S can vary in comparison to other genes when analyzed by sensitive Real-Time PCR techniques (Bustin 2000, Akilesh et al., 2003). Any small variation in the normalizer amplification would therefore compromise the analysis of the complete Real-Time PCR data set. Real-time dogma #2 - ranking genes
strictly by fold change
When a single gene normalizer is selected, gene expression changes are typically ranked by their magnitude of change using the ΔΔCt method, with those genes showing the largest fold changes ranked as most significant. Unfortunately, these large changes in gene expression may mask small, but biologically important changes in gene expression, such as master regulator genes (e.g., transcription factors). In biology, however, larger is not always synonymous with importance. To combat the above mentioned problems, Bar Harbor Biotechnology, Inc. developed a modified Global Pattern Recognition™ algorithm (Akilesh et al., 2003), which is optimally suited to generate a ranked list of significantly changed genes within a Real-Time PCR dataset. This unique algorithm and accompanying software overcomes the problem of identifying invariant normalizers and the pitfalls of producing faulty statistics based merely on magnitude of change. Global Pattern Recognition provides a true statistical analysis of results based on consistency in the data, which makes Global Pattern Recognition™ optimally suited to detect small, but reproducible changes. Only after the genes are statistically ranked is the magnitude of the change calculated. A typical experiment would utilize 'biological replicates' (Bio-Reps). Bio-Reps are defined as samples collected from separate and closely matched biological samples. They are processed individually under closely matched conditions. Advisedly, it is best to analyze at least 3 bio-reps in each of two groups, representing for example '3 sick vs. 3 healthy' or '3 treated vs. 3 untreated' groups (but Global Pattern Recognition can handle much larger data sets). Global Pattern Recognition processes the data derived from groups of Bio-Reps and reveals the 'constellation' of changing genes. Each constellation can be evaluated for the most likely biological context providing the researcher with a better understanding of the overall results. Just as early sea navigators used the stars to triangulate their position on the ocean, Global Pattern Recognition globally positions the expression level of each gene with respect to all genes within an experiment. This can be done without prior assumption that a gene (normalizer) has an invariant expression level. Global Pattern Recognition is unbiased in that it enables the experimental data to define the invariant normalizer genes, not the experimenter. The use of any gene as a potential normalizer also maximizes the use of the limited real-estate on a StellARray™ plate by eliminating the loss of wells used to contain potentially erroneously predefined normalizers. Global Pattern Recognition is extremely simple to use and reliably tabulates statistical significance (p-value) of gene expression changes on the fly allowing you to immediately focus on the real biology. Simply log into GPR, select the StellARray that you ran on your Real-Time PCR instrument, upload your data and submit for analysis. An HTML or Excel® formatted file will be generated that gives a ranked list of genes by p-value, fold change value, and links to MGI and NCBI gene pages. With each purchase of a StellARray™ pack your account will be receive analysis query credits sufficient to analyze each plate.
SA Biosciences (a QIAGEN company) SABiosciences
leads the field in high-performance SYBR Green
real-time PCR analysis. Our RT² Profiler PCR
Arrays (patent pending) analyze expression of a
panel of genes associated with any one of over 100
biological pathways or specific disease states.
The RT² qPCR primer Assays are experimentally
tested and validated qRT-PCR primer sets for every
human, mouse, rat, rhesus macaque, or even fruit
fly gene. These qPCR primer assays are
ready-to-use for gene-by-gene expression analysis,
microarray data validation, biomarker discovery
and siRNA knock-down verification. In combination
with our special formulated and
instrument-specific PCR master mixes, our complete
RT² qPCR products provide the accurate, reliable,
and convenient SYBR Green analysis needed by
today's research. Trust the experts in SYBR Green
real-time PCR detection.
Genome-wide microRNA detection by real-time PCR => Download White Paper
Roche Applied Science
Exiqon
Life Technologies™ products Custom TaqMan® Assay Manufacturing & Plating Service If your research application requires special manufacturing modifications - such as a different dye or assay volume, or you would like to have your assays plated in a specific way, our custom services can help. TaqMan® Gene Expression Assays TaqMan® Gene Expression Assays provide over 1.3 million predesigned primer/probe sets covering 23 species, the most comprehensive set of quantitative gene expression assays available. Alternatively, custom assays enable you to study the expression of any gene or splice variant in any organism. TaqMan® MicroRNA Assays Innovative TaqMan® Assays for microRNA and other small RNAs, as well as longer noncoding RNA transcripts such as pri-miRNAs and long noncoding RNA. We offer products for noncoding RNA discovery, profiling, quantitation, validation, and functional analysis. TaqMan® Assays for Genotyping & Genetic Variation Analysis The precision of TaqMan® probe-based chemistry makes SNP genotyping and copy number variation studies more accurate than ever. Choose from 4.5 million human and mouse TaqMan® SNP Genotyping Assays or design your own Custom TaqMan® SNP Genotyping Assays. For analysis of SNPs in genes for drug metabolizing enzymes (DMEs), we offer 2,700 unique TaqMan® DME Assays that detect polymorphisms in 221 genes for DMEs and associated transporter targets. For copy number variation studies, we offer over 1.6 million predesigned TaqMan® Copy Number Assays with human, genome-wide coverage, and mouse and marker/reporter assays. Custom and Custom Plus TaqMan® Copy Number Assay options are ideal when a predesigned assay for a target is not available.
TaqMan® Protein Assays Revolutionary TaqMan® Protein Assays enable fast, easy identification, and relative quantification of protein markers from limited quantities of cultured cells. Choose from predesigned assays for human stem cell pluripotency markers and control proteins, or create your own assay from your biotinylated antibodies. WaferGen Next Generation Real-time PCR Comes of Age The WaferGen SmartChip System enables profiling and validation workflows on a single platform, by combining high-throughput, cost-effective target discovery with the sensitivity, precision, and dynamic range of real-time PCR for validation studies. Gene Expression Profiling - Discover More Gene expression profiling of specific diseases has become increasingly important in drug development. Comparison of gene expression patterns between normal and diseased patients or expression profiles in the presence or absence of drugs leads to discovery of genes or a set of genes that can be used in drug development. This requires monitoring of tens, hundreds or thousands of mRNAs in large numbers. The WaferGen SmartChip System offers the capability to achieve this level of throughput with a high degree of accuracy. MicroRNA Profiling - Comprehensive human microRNA profile on a single chip MicroRNAs (miRNA) are post-transcriptional regulators of cell proliferation, tissue differentiation, embryonic development, and apoptosis. Specific miRNA expression profiles may be characteristic of diseases or disease states and used as biomarkers. The identification of miRNA profiles has become important for streamlining drug development processes. The comparison of miRNA patterns from normal and disease samples; or contrasting miRNA profiles of the same sample in the presence or absence of a drug leads to a greater understanding of pathways and mechanisms of action. The WaferGen SmartChip System in conjunction with the pre-validated SmartChip human miRNA panel, offers researchers the ability to carry out comprehensive, rapid miRNA profiling on their human samples simply, cost-effectively, and accurately. Fluidigm Fluidigm’s
BioMark 96x96 Dynamic Arrays These
new
integrated fluidic circuits (IFCs) are capable of
performing 9,216 simultaneous experiments without
any pre-spotting and with complete flexibility for
the researcher. In a word, they are
“revolutionary.” Flyer The 96.96 Dynamic Arrays are at the heart of Fluidigm’s BioMark™ Genetic Analysis System, which delivers new efficiencies in Gene Expression and Genotyping. Fluidigm products enable and accelerate your Single Cell Gene Expression, Copy Number Variation and Absolute Quantitation research in completely new ways. RealTimePrimers.com Library of
targeted real time PCR primer sets
Perform up to 200 PCR Arrays! Flexibility to design one's own experiments Microplate containing 88 targeted plus 8 housekeeping gene primer sets Note: Primer sequences unavailable for primer libraries
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||